Apply for and Manage a Biohazard Use Authorization (BUA) for Laboratory Research or Clinical Trials Using Biohazardous Materials
Apply for and Manage a Biohazard Use Authorization (BUA) for Laboratory or Clinical Research Using Biohazardous Materials.
Access BUA on the My Research Safety Web portal.
Principal investigators must obtain approval from the UC San Diego Institutional Biosafety Committee (IBC) before beginning research, clinical, or teaching activities with biohazardous materials or recombinant or synthetic nucleic acids. The UC San Diego IBC (Camus IBC) and UC San Diego Human Gene Therapy IBC (HGT IBC) utilize the online Biohazard Use Authorization (BUA) application.
An approved BUA will:
- Describe the work to be conducted with biohazardous materials
- Establish the biosafety level of containment
- Identify the researchers, lab workers, and collaborators
- Authorize the PI to conduct the work
- Identify the approved work locations
PIs are responsible for initiating a BUA, keeping their BUA current at all times, and submitting a BUA for IBC review. Note, PI Contacts designated within the BUA application are eligible to view, add personnel, and make edits to a BUA application under draft status
UC San Diego IBC holds virtually meetings on the second Friday of the month from 9–11 a.m. For any questions regarding the IBC and scheduled meetings, contact us.
UC San Diego Human Gene Therapy IBC holds meetings on the second Tuesday of the month. For information regarding submitting a clinical BUA and questions regarding the meetings, visit Clinical Trials with rDNA.
Overview
IBC review
Comprehensive review by the IBC is made of all the biohazardous work proposed in any given laboratory or clinical setting.
The combination of all work proposed by a research group—regardless of the source of funding (or even lack of funding)—is subject to a comprehensive review. Therefore, for the purposes of the BUA review, a principal investigator* shall be the ultimate, single point of accountability and responsibility for a given laboratory or clinical trial.
Campus IBC reviews research activities that involve biohazardous materials, human source materials, environmental samples, recombinant or synthetic nucleic acids, etc. that are unfunded, federally funded, PI-funded or received approval by the FDA for compassionate use via an emergency authorization (e.g., expanded access program, single patient IND). Research cannot be initiated until the IBC has approved the protocol.
Human Gene Therapy IBC
The Human Gene Therapy IBC reviews research activities that involve administration of recombinant or synthetic nucleic acid molecules as defined under the NIH Guidelines III-C-1 into human research participants. As part of the service, Sabai Global will initiate, submit, and maintain the BUA on behalf of the PI. Contact Sabai Global at clientservices@sabaiglobal.com to initiate the submission and review process.
*Principal Investigator is defined as a person who is responsible for the research project and all those involved and is assigned laboratory space or is conducting a human gene transfer clinical trial.
Related research authorizations
Other research authorizations for work with regulated materials or human or animal subjects at UCSD may be required.
See Research Safety Authorizations for Principal Investigators.
BUA application
The Biohazard Use Authorization (BUA) application provides a standardized, secure, and web-based program for managing research, clinical, and teaching activities involving biohazardous materials or recombinant or synthetic nucleic acids.
Use the BUA application to apply for a new BUA, and to amend or renew an active authorization. For industry-funded clinical trials, contact Sabai Global at clientservices@sabaiglobal.com
Approval must be sought and obtained from the IBC prior to the initiation of work involving any of the following (this list is not inclusive):
- New infectious agents
- New viruses, new vector systems
- Enhanced replication or infectivity of ;viruses of microorganisms
- Biological toxins
- Work with human or primate cell lines
- Nucleic acids
- Changes in genetic constructs or model systems
- Procedures with increased risk due to aerosolization or another type of exposure to biohazards
- Any recombinant nucleic acids used in vitro, in vivo, and in clinical trials
Submission dates
BUA applications are reviewed at the IBC's monthly meeting.
See IBC Meeting and Submittal Dates (PDF) for due dates. For any questions regarding the IBC and scheduled meetings, email ehsbio@ucsd.edu.
- Due to the extensive pre-review process, we strictly adhere to the deadlines.
- Exceptions to submission and meeting dates can be made in cases of emergency use authorizations or expanded access involving patients. In these cases, contact EHSBio@ucsd.edu and request an expedited review.
Life cycle of a BUA application
The Biohazard Use Authorization (BUA) Application follows a structured review and approval process to ensure that biohazardous materials are used safely, compliantly, and under appropriate institutional oversight. Below are the key stages in the life of a BUA, from initiation to final approval.
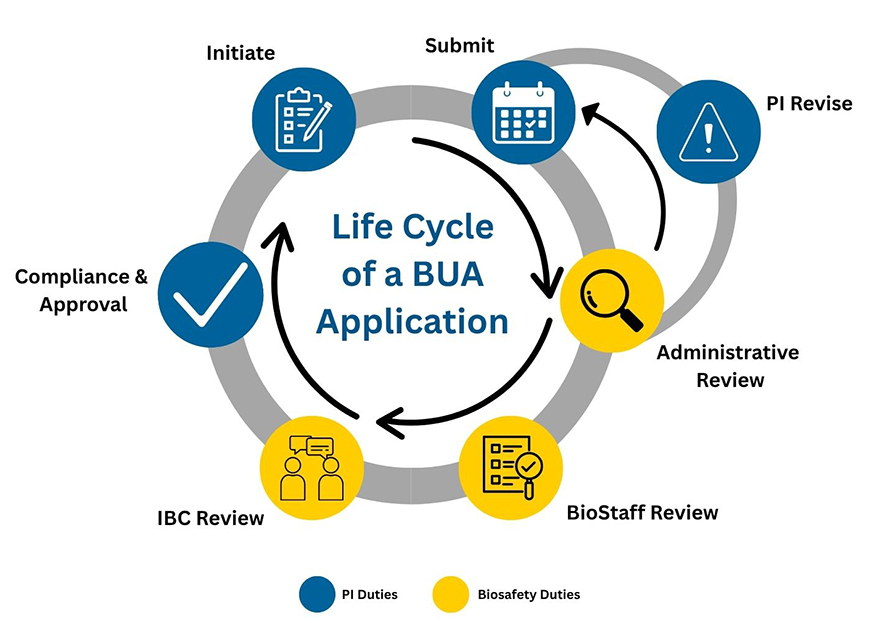
1. Initiation of BUA
The BUA life cycle begins when a Principal Investigator (PI) initiates a new application. Labs initiate a BUA from the home page or under the tab My BUAs by selecting the needed task. Here are the options:
- New BUA
- Renew
- Amend
Note: Before selecting the Renewal or Amendment action button, select the radio button for the BUA you wish to Renew or Amend.
Key characteristics:
- A new BUA record is created in the system.
- The application is saved in Draft
- Project details (e.g., personnel, materials, procedures, locations) are not yet complete. A red X next to a section within the BUA application indicates the section has not been completed.
- The application is editable by the PI and the designated PI Contact(s) indicated in Section 9 of the BUA.
BUA Application Status for Initiated BUA:
The Request Type column will list Draft until submitted and the Review Stage column will list N/A for new, amend, or renewal applications that are initiated and in progress for editing by the PI or PI Contact. Within the BUA application under the My BUAs tab, the BUA listing will show an initiated BUA as the following Request Type, Request Status, and Review Stage:
- Request Type: Draft
- Request Status: Blank
- Review Stage: N/A
Outcome: A New BUA will be assigned a BUA number. For initiation of Amendments or Renewals, the BUA number remains the same and the Request Type changes from New to Amend or Renew.
2. Complete Content and Submit
The PI and/or PI Contact completes all required sections of the BUA and the PI formally submits the application for review.
Key characteristics:
- All mandatory fields and applicable Hazard Control Plans are completed.
- Personnel training and roles are identified.
- Biohazard materials and containment practices are described.
- The PI certifies the accuracy of the information.
- The application is locked for editing upon submission.
BUA Application Status for Submitted BUA:
The Request Type column will identify the type of protocol, either New, Amend, or Renew. The Request Status column will update to submitted and the Review Stage column will list Admin for submitted applications. At this stage, the PI or PI contact can no longer make edits to the application.
- Request Type: New, Amend, or Renew
- Request Status: Submitted
- Review Stage: Admin
Outcome: The BUA enters the administrative review workflow.
3. Post-Submission: Administrative Review
Administrative staff conduct an initial screening to ensure the application is complete and ready for scientific and safety review.
Key characteristics:
- Review for completeness, formatting, and required documentation.
- Verification of personnel eligibility and training records.
- Identification of missing or unclear information.
BUA Application Status for BUAs under Administrative Review:
The Request Type column will identify the type of protocol, either New, Amend, or Renew. The Request Status column will remain as submitted and the Review Stage column will list Admin.
- Request Type: New, Amend, or Renew
- Request Status: Submitted
- Review Stage: Admin
Possible actions:
- Return to Principal Investigator for corrections or clarifications. The BUA will be placed under PI Revise. This allows the research team to make corrections and clarifications and, once complete, resubmit the BUA.
- Advance to BioStaff Review if administratively complete.
Outcome: The BUA is either revised by the applicant or forwarded for technical review.
4. BioStaff Review
Biosafety professionals review the application for technical accuracy and compliance with biosafety and regulatory standards. Additionally, the biosafety staff will perform a risk assessment and contact the PI and PI Contact for additional information and/or to conduct a site visit.
Key characteristics:
- Evaluation of biohazard risk level and containment.
- Assessment of laboratory practices, equipment, and facilities.
- Review of decontamination, waste disposal, and emergency procedures.
BUA Application Status for BUAs under Biostaff Review:
The Request Type column will identify the type of protocol, either New, Amend, or Renew. The Request Status column will remain as submitted and the Review Stage column will list Biostaff.
- Request Type: New, Amend, or Renew
- Request Status: Submitted
- Review Stage: Biostaff
Possible actions:
- Request revisions or additional information.
- Recommend approval to the Institutional Biosafety Committee (IBC).
Outcome: The BUA is prepared for committee-level review.
In some cases where the research meets the requirements for low-risk, the BUA is discussed, reviewed, and approved by the Biostaff and acknowledged at a convened IBC meeting following the designated review process. Note, designated review does not apply to research that falls under the NIH Guidelines III-A to III-E.
5. IBC Review
The Institutional Biosafety Committee (IBC) reviews the application to ensure regulatory and institutional compliance.
Key characteristics:
- Committee members review risk assessments and mitigation strategies.
- Discussion must occur during a convened meeting for research involving recombinant or synthetic nucleic acid molecules that falls under the NIH Guidelines III-A to III-E. Discussion may occur through a designated review process for low-risk research that does not involve recombinant or synthetic nucleic acids.
- Additional stipulations or modifications may be required.
BUA Application Status for BUAs under IBC Review:
The Request Type column will identify the type of protocol, either New, Amend, or Renew. The Request Status column will update to submitted and the Review Stage column will list IBC or Primary Review.
- Request Type: New, Amend, or Renew
- Request Status: Submitted
- Review Stage: IBC or Primary Reviewer
Possible decisions:
- Approved
- Acknowledge Approval of Designated Reviewed BUAs
- Approve Pending completion of itemized tasks
- Defer to another IBC meeting
- Not approved
Outcome: A formal determination is issued.
For Approve Pending or Deferred, a formal determination is sent to the lab for a response within 60 days. After 60 days, if no response has been provided by the lab, the protocol will be Not Approved and the lab will need to start the BUA application process over. Exemptions to the 60-days can be granted on a case by case basis.
6. Compliance and Approval
Once all required reviews are successfully completed, the BUA is approved. For Approvals, the lab will receive a formal IBC Approval Letter that details any approval conditions. Approval conditions must be implemented prior to start of work.
Key characteristics:
- The Approval status changes to Approved.
- The Principal Investigator and PI Contacts will receive an email with the formal IBC Approval Letter. The Letter can also be found within the BUA Application under Section 8. Attachments and Section 11. BUA Reports.
- Research activities involving biohazardous materials may begin.
- Approval is valid for a defined period, subject to renewals and amendments.
- The BUA is subject to ongoing compliance monitoring.
BUA Application Status for Approved BUAs:
The Request Type column will identify the type of protocol, either New, Amend, or Renew. The Request Status column will update to submitted and the Review Stage column will list Complete. At this stage, the lab has met all approval conditions and received an IBC Approval Letter. BUA Amendments and Renewal can be initiated once an IBC Approval Letter is received by the lab.
- Request Type: New, Amend, or Renew
- Request Status: Submitted
- Review Stage: Completed
Outcome: The BUA enters its active lifecycle phase, supporting safe and compliant research operations.
The PI is responsible for updating the BUA with applicable information via an Amendment or Renewal.
How to apply for a BUA
Step 1: First-time applicants
Two things must happen before you can access the BUA application to apply for a Biohazard Use Authorization:
- You must be entered in the Environment, Health & Safety (EH&S) database to enable certain research safety authorizations and access EH&S online applications. Use the PI Information form to be entered into the EH&S database or to report information changes.
- You must have a UCSD Active Directory or a UCSD Business Systems (sometimes referred to as Single Sign-On) username and password to access the BUA application. See How to Get Access to UC San Diego Business Systems for simple instructions. Contact your departmental security administrator (DSA) if you need assistance.
Step 2: Apply for a BUA
- Go to My Research Safety Web portal.
- Click the Authorizations tab and select to open the application.
- Follow instructions on the welcome screen to apply for a new BUA.
Choose the appropriate type of BUA. Two types of BUA applications are offered:
- Type 1: Research BUA – for standard research
- Type 2: Clinical Trial BUA– for human gene transfer (gene therapy).
It is recommended that Principal Investigators designate a PI Contact within Section 9 of the BUA application. PI Contacts can have 1 of 2 status:
- PI Contact with editing privileges
- PI Contact with view only privileges
PI Contact with editing privileges can:
- Make edits to an initiated New, Amendment, or Renewal BUA
- Add/Remove personnel at any time from the BUA
- View the BUA
- Receive emails regarding the status of the BUA, personnel training requirements, and communications during the administrative and Biostaff reviews.
PI Contact with view only privileges can:
- View the BUA
- Receive emails regarding the status of the BUA, personnel training requirements, and communications during the administrative and Biostaff reviews.
Note: For PIs with multiple BUAs, the PI Contacts remain the same for all BUAs.
Amend your BUA
When to amend your BUA
The PI must notify EH&S and obtain IBC approval via the BUA amendment process before changing any variable which may carry increased biohazard risk. Notification and subsequent IBC approval are also required of the PI before any given COMBINATION of such variables may be changed.
As a working principle, PIs must decide if a new agent or protocol poses any new or increased hazards from those indicated in the currently approved proposal. If there are new or increased hazards (or if there is any doubt or question), approval must be sought prior to implementing the change.
The PI must submit an amendment for
- Addition/Deletion of Locations
- Addition/Deletion of Biological Materials
- Changes in genetic constructs
- Changes in SOP
- Changes in participant enrollment for Clinical trial BUA
- Updates in Serious Adverse Event (SAE) for clinical trial BUA
Amendments require IBC approval before the new work can commence.
Use the BUA (Biohazard Use Authorization) application to amend your BUA.
How to amend your BUA
- Go to My Research Safety.
- Click the Authorizations tab and select to open the application.
- Select your BUA from the My BUAs tab and press the Amend function button at the bottom of the screen to proceed.
How to write an Amendment Project Summary
Amendments require a project summary describing what changes are being made to the BUA. An amendment summary should include the following elements:
- Include a project description for added projects or agents
- Include a description and identify the project for any materials or projects that were removed
- Write what experimental procedures and assays you plan to perform
- Detail the risks and any risk mitigation measures in place
Renew a BUA
BUAs must be renewed every 1, 3, or 5 years, depending on the risk. PI’s are responsible for submitting a renewal to the BUA prior to the renewal date.
The renewal date is available on the approval letter and within the BUA application. A renewal notice is emailed to the PI and PI Contact starting 2 months before the renewal date.
To encourage early submission, BUAs submitted more than 2 months in advance, the IBC will add time to the next renewal cycle based on how early the BUA is renewed, up to a maximum of 6 months. For example, if a 3-year renewal BUA is submitted 4 months prior to the renewal date, the IBC will approve the next renewal cycle to be 3 years and 4 months.
Note, the IBC allows previously approved research to continue during the renewal process. New work described on a BUA Renewal cannot be initiated until IBC approval.
BUAs must be submitted prior to the renewal date. For BUAs that are submitted after the renewal date, research cannot occur until an active IBC Approval is issued.
How to write a Renewal Project Summary
Renewals require a rewrite describing the current scope of work occurring in the lab.
For renewal with changes, update the following within the project summary and the applicable sections of the application:
- Add new projects and agents,
- Move agents/materials into storage
- Remove projects that are no longer performed by the lab and identify what was removed.
If the lab is submitting a renewal where no changes have occurred, identify that no changes have occurred and provide a rewrite of the project summary.
Renewals that do not include a rewrite of the project summary will be reviewed during the Admin stage and returned through PI Revise for the lab to include the rewrite.
Update personnel or training anytime
The BUA application allows a PI or PI Contact to do the following without creating or submitting an amendment
- Add or remove authorized personnel – Note: lab members must first be listed on the My EH&S Profile – Lab Roster before being added into the BUA application.
- Add or remove PI contacts and assign editing privileges
- Assign required biosafety training based on personnel lab assignments.
Authorized personnel can enroll for required training using UC Learning Center. See required biosafety training.
Request a BUA verification letter
Researchers may need to submit biosafety assurances to funding agencies. Many funding agencies accept the IBC approval letter for grants that cover projects listed on an approved BUA. However, there are instances when a grant requires specific language on a biosafety assurance.
Researchers can request a BUA Verification Letter that can be sent to funding agencies with the required language and proof of institutional oversight for the use of recombinant DNA, infectious agents, etc. and assurance that the applicant is compliant with regulations applicable to use of biohazardous materials.
How to get a BUA verification letter:
- Complete a BUA Verification Letter for Funding Agencies request form.
- A verification letter signed by the UCSD Biosafety Officer will be sent to you according to instructions you provide on the request form.
Important: If the request is urgent please include "URGENT" in the subject line of your email.
Deactivate a BUA
Deactivate a BUA when the responsible PI retires, leaves or no longer possesses UC San Diego regulated biological materials. Closing out authorizations should be completed prior to leaving the institution.
A BUA can be deactivated and closed only if:
- All activity has stopped,
- All biological materials have been removed and disposed of, and
- All work surfaces and equipment have been decontaminated.
The biosafety program requires PI’s to verify and attest that all three of the requirements have been met before a BUA is deactivated.
How to deactivate a BUA:
Request BUA deactivation by notifying EH&S Biosafety in writing:
- Reference the BUA Number and PI's name.
- Include a short description of how biohazardous agents and materials were removed from the lab.
- For Clinical Trials: confirm requirements for closeout (see below)
Notify EH&S Biosafety by email: EH&S Biosafety
Clinical trial BUAs
Clinical trial BUA’s can be deactivated once the project meets all of the following criteria:
- It has been one month since the last dosing of a participant.
- All investigational new drugs have been destroyed or returned to sponsor.
- Study is closed to enrollment.
Transfer a BUA
There are instances when a project or an entire BUA is now overseen by a different PI. In these cases, the application must reflect the change. PIs that are taking over 1) entire BUA or 2) specific projects and materials will need to create a new BUA or add the information to their already existing BUA.
How to transfer a BUA:
- The receiving Principal Investigator (PI) should start a new BUA application in the My Research Safety portal (see How to Apply for a BUA).
- In the Project Summary field, note that this is a BUA transfer and list:
- The previous BUA number and PI’s name
- A description of the biological material(s) and procedure(s) that will be transferred
- Then add the new PI (or PI Contact) as authorized personnel on the original BUA (the one to be transferred). They will then be able to copy over the relevant information from the previous BUA protocol to the new BUA protocol.
Any agents that have already been disposed of or transferred to another location do not need to be listed again.
How to transfer biological materials between BUAs:
- The receiving Principal Investigator (PI) must add newly acquired materials into their BUA using the My Research Safety portal (see How to Apply for a BUA). PI’s can initiate an Amendment to add materials.
- In the Project Summary field, add the following:
- A description of the biological material(s) and procedure(s) that will be transferred
- Then add the new PI (or PI Contact) as authorized personnel on the original BUA (the one to be transferred). They will then be able to copy over the relevant information from the previous BUA protocol to the new BUA protocol.
Related links: See How to Close or Relocate a UC San Diego Laboratory if the lab is being closed.
Managing Hazard Control Plans (HCP) and BUAs
This tutorial provides an introduction step-by-step instruction for how to create and manage Hazard Control Plans (HCP) and BUAs:
(Note: If the embedded video does not play, try opening page in incognito mode and/or delete your cookies/cache history.)
